

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133193 (CHEMBL337721 | Neurotensin analogue) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.230 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124132 (CHEMBL3622802) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.260 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133188 (CHEMBL336836 | Compound KK17) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50342242 ((S)-2-((2S,3S)-2-((S)-2-((S)-1-((S)-6-amino-2-((S)...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | n/a | n/a | 0.470 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133198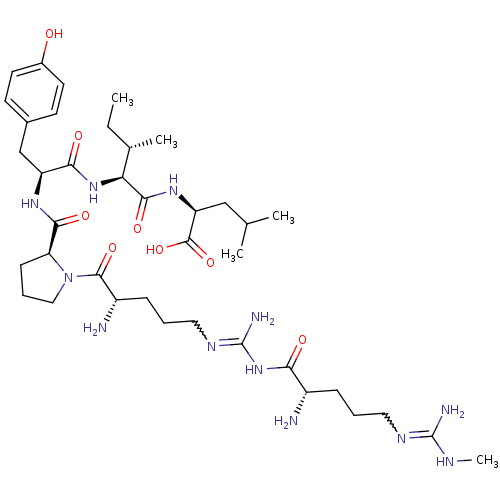 (CHEMBL434227 | Compound KK16) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.670 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133206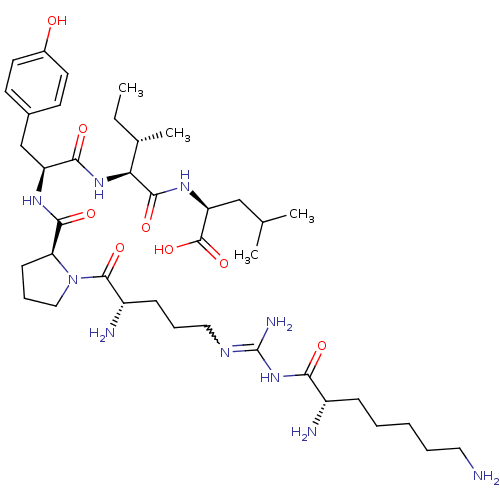 (CHEMBL337644 | Compound KK1) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.700 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133201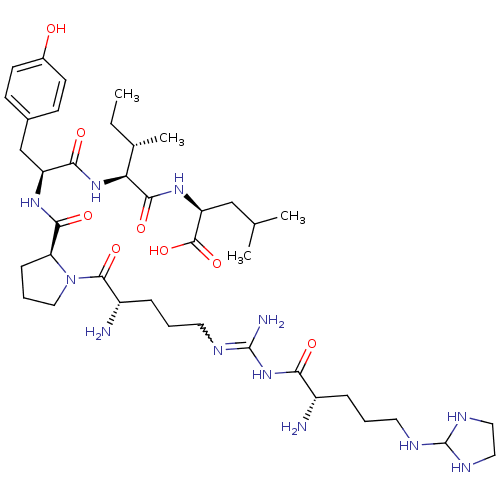 (CHEMBL132524 | Compound KK18) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.780 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133204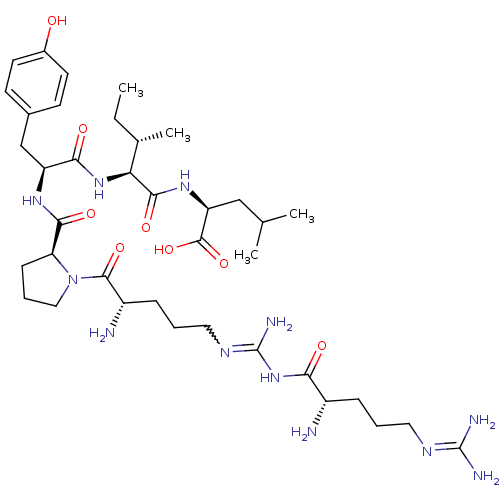 (CHEMBL132409 | Compound KK15) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50342242 ((S)-2-((2S,3S)-2-((S)-2-((S)-1-((S)-6-amino-2-((S)...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.950 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of 125I-[Tyr3]-NT from human NTS2 receptor | J Med Chem 60: 3303-3313 (2017) Article DOI: 10.1021/acs.jmedchem.6b01848 BindingDB Entry DOI: 10.7270/Q2377C6V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50130880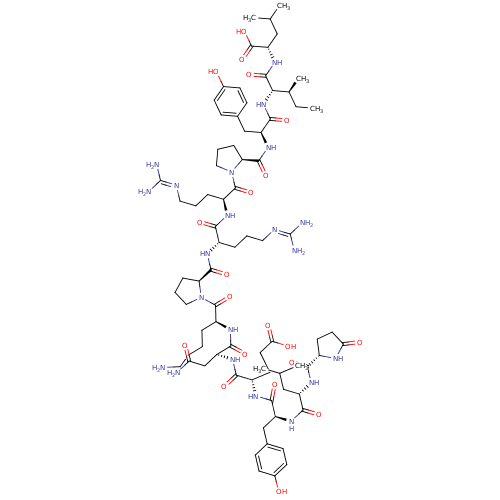 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | n/a | n/a | 1.10 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133196 (CHEMBL337260 | Compound KK6) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133190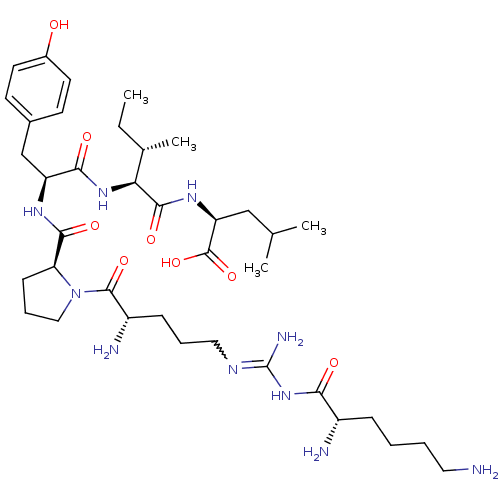 (CHEMBL430910 | Compound KK5) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50240845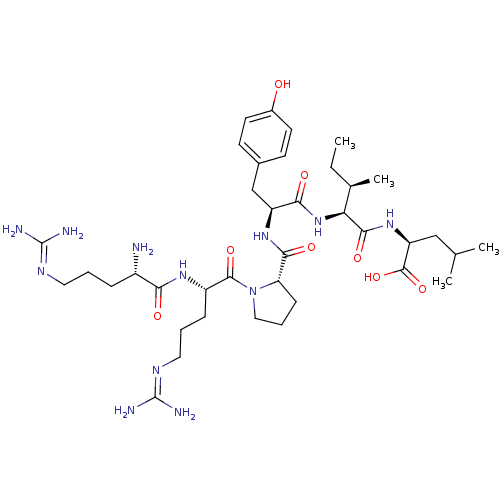 ((S)-2-{(2S,3R)-2-[(S)-2-({(S)-1-[(S)-2-((S)-2-Amin...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133191 (CHEMBL132507 | Compound KK7) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.80 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133199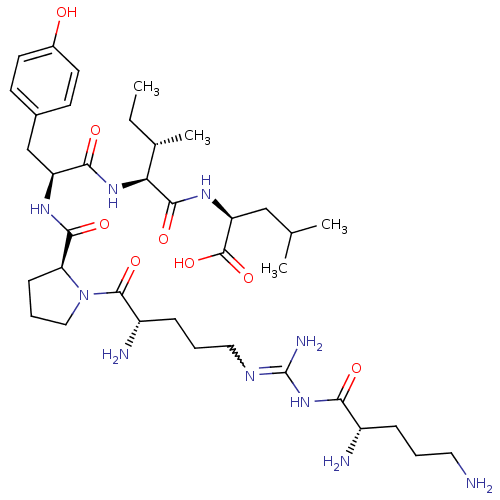 (CHEMBL339139 | Compound KK9) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133202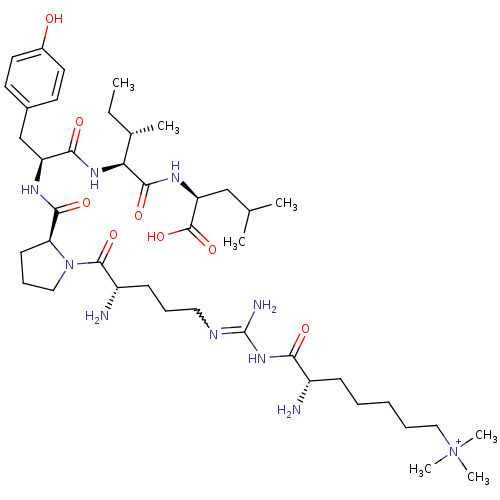 (CHEMBL132794 | Compound KK4) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133205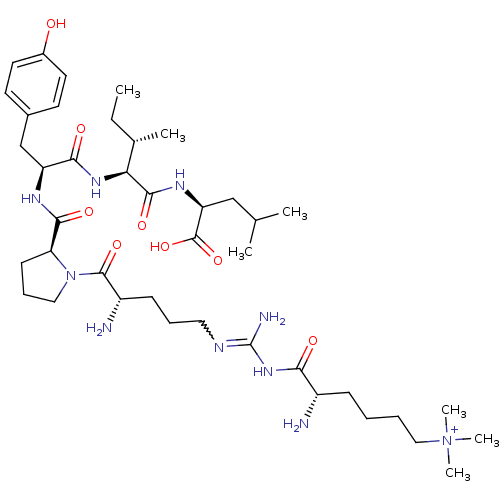 (CHEMBL133798 | Compound KK8) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.20 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133195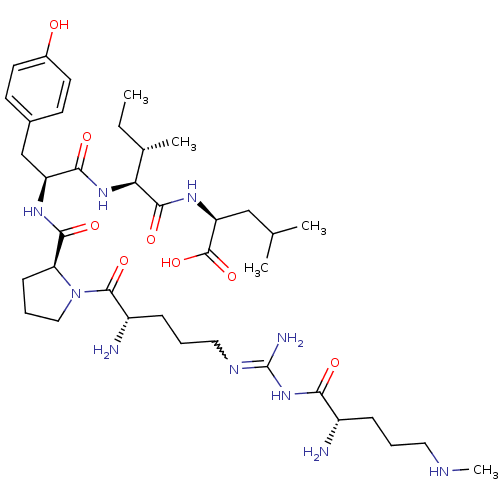 (CHEMBL133340 | Compound KK10) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133187 (CHEMBL133850 | Compound KK3) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133192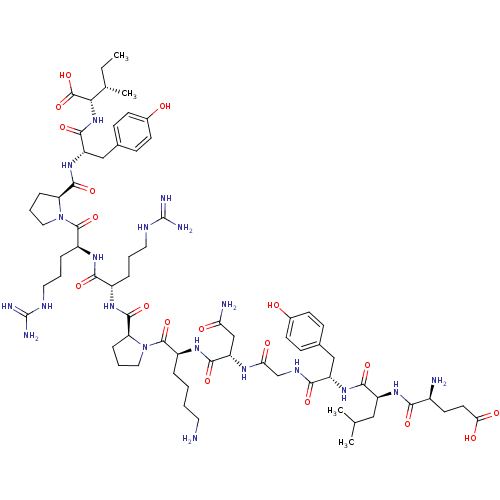 (CHEMBL439494 | Compound NT) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124143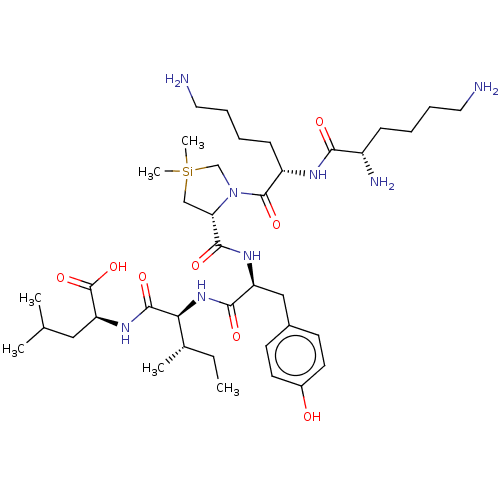 (CHEMBL3622806) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a | |
TBA | Citation and Details | ||||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133207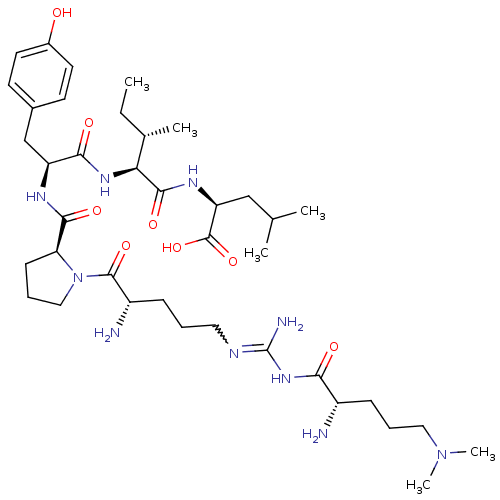 (CHEMBL133378 | Compound KK11) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133200 (CHEMBL129953 | Compound KK12) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50240845 ((S)-2-{(2S,3R)-2-[(S)-2-({(S)-1-[(S)-2-((S)-2-Amin...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 stably expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133194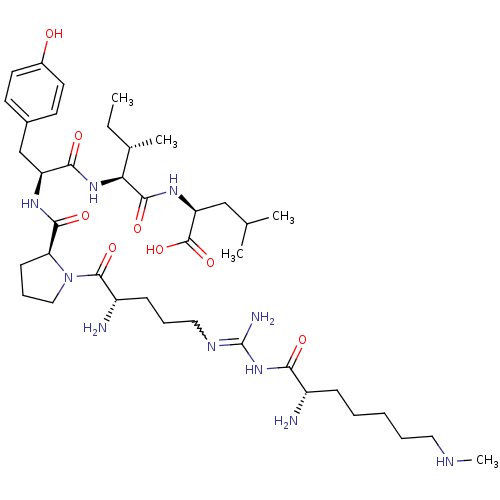 (CHEMBL133849 | Compound KK2) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50048908 (CHEMBL415788) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 7.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133197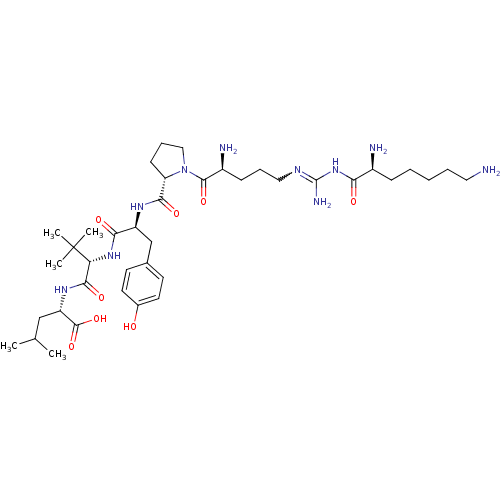 (CHEMBL335846 | Compound KK13) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 11.1 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 stably expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124143 (CHEMBL3622806) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124135 (CHEMBL3622803) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 29 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124141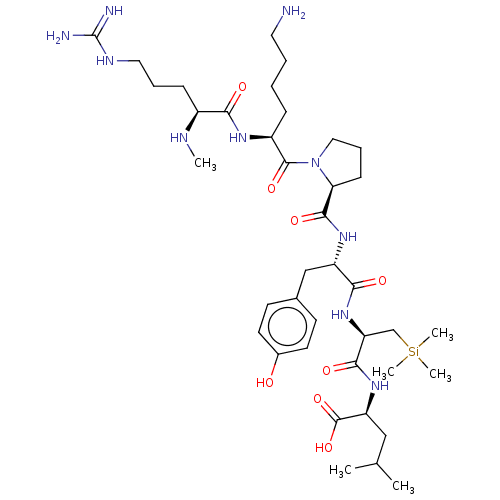 (CHEMBL3622805) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50023628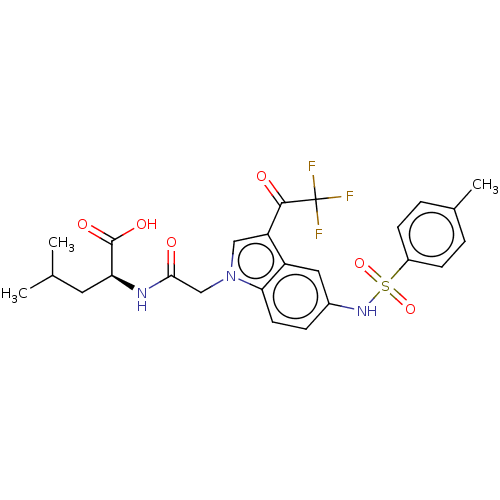 (CHEMBL3326831) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 38 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 receptor expressed in CHO-K1 cells assessed as inhibition of SR142948a-induced calcium mobilization by FLIPR assay | J Med Chem 57: 7472-7 (2014) Article DOI: 10.1021/jm500857r BindingDB Entry DOI: 10.7270/Q2BP04C6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 42 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat neurotensin receptor type 2 expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR ... | Bioorg Med Chem Lett 25: 292-6 (2014) Article DOI: 10.1016/j.bmcl.2014.11.047 BindingDB Entry DOI: 10.7270/Q2M90B9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50130880 (CHEMBL407196 | NT(1-13) | neurotensin | pGlu-Leu-T...) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 114 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 receptor expressed in CHO-K1 cells assessed as inhibition of SR142948a-induced calcium mobilization by FLIPR assay | J Med Chem 57: 7472-7 (2014) Article DOI: 10.1021/jm500857r BindingDB Entry DOI: 10.7270/Q2BP04C6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50019423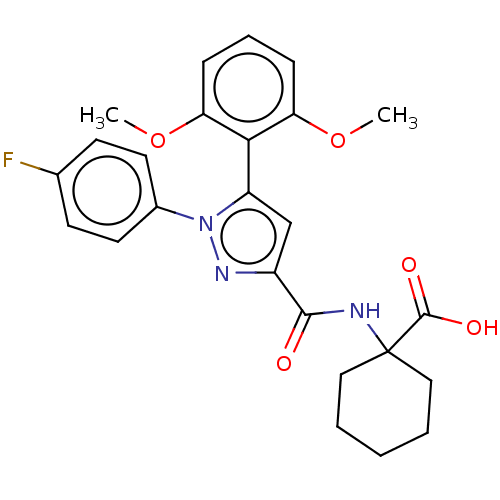 (CHEMBL3290105) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 140 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 stably expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50019424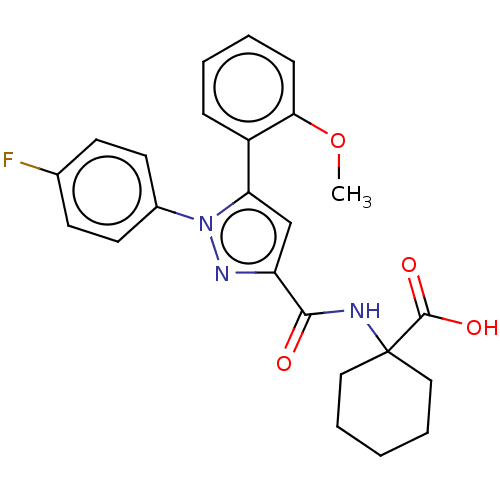 (CHEMBL3290106) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 153 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 stably expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR assay | J Med Chem 57: 5318-32 (2014) Article DOI: 10.1021/jm5003843 BindingDB Entry DOI: 10.7270/Q2PR7XJD | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124146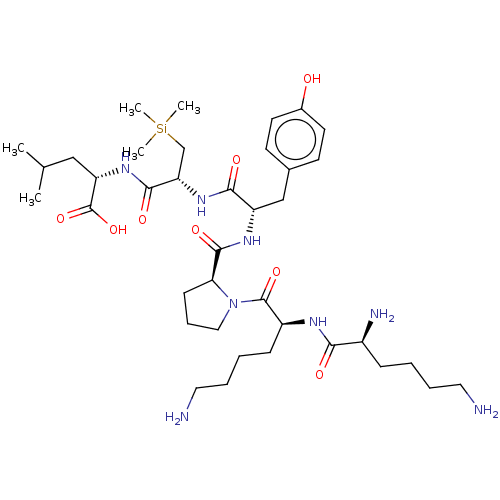 (CHEMBL3622801) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 405 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50133189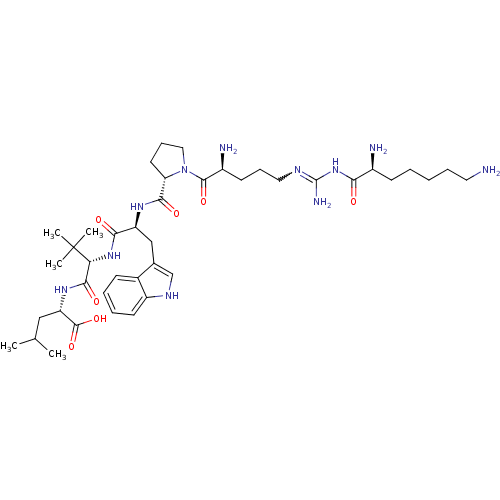 (CHEMBL435199 | Compound KK14) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 500 | n/a | n/a | n/a | n/a | n/a | n/a |
Medical University of South Carolina Curated by ChEMBL | Assay Description Evaluated for binding affinity by inhibiting binding of [125I]Tyr(3)-NT to human Neurotensin receptor 2 | J Med Chem 46: 4141-8 (2003) Article DOI: 10.1021/jm0300633 BindingDB Entry DOI: 10.7270/Q26M37KB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50041428 (CHEMBL3356855) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.07E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat neurotensin receptor type 2 expressed in CHOK1 cells assessed as inhibition of SR142948a-induced calcium release by FLIPR ... | Bioorg Med Chem Lett 25: 292-6 (2014) Article DOI: 10.1016/j.bmcl.2014.11.047 BindingDB Entry DOI: 10.7270/Q2M90B9G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Homo sapiens (Human)) | BDBM50124138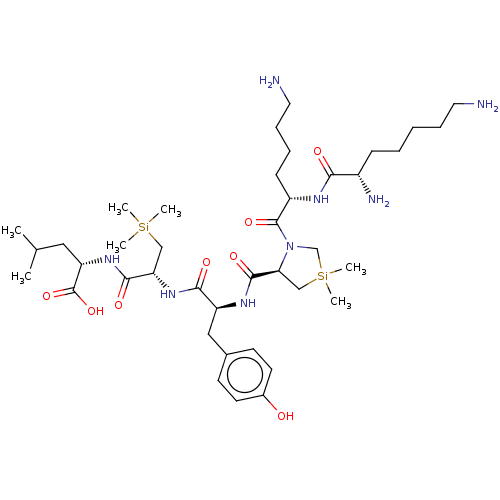 (CHEMBL3622804) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.43E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ Montpellier Curated by ChEMBL | Assay Description Displacement of [125I]-Tyr3-NT from human NTS2 receptor expressed in 1321N1 cell membranes incubated for 30 mins by gamma-counting based competitive ... | J Med Chem 58: 7785-95 (2015) Article DOI: 10.1021/acs.jmedchem.5b00841 BindingDB Entry DOI: 10.7270/Q2MW2JZ3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neurotensin receptor type 2 (Rattus norvegicus) | BDBM50023627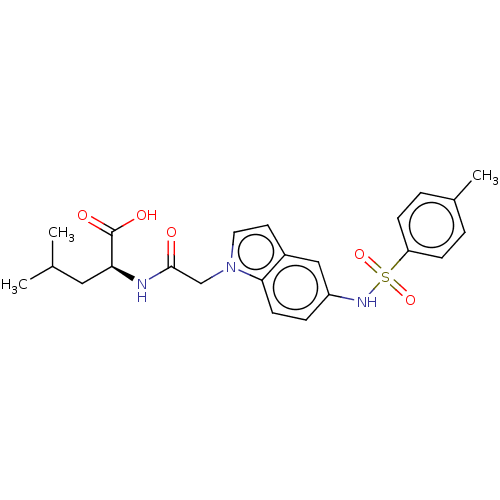 (CHEMBL3326832) | KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 3.32E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Research Triangle Institute Curated by ChEMBL | Assay Description Antagonist activity at rat NTS2 receptor expressed in CHO-K1 cells assessed as inhibition of SR142948a-induced calcium mobilization by FLIPR assay | J Med Chem 57: 7472-7 (2014) Article DOI: 10.1021/jm500857r BindingDB Entry DOI: 10.7270/Q2BP04C6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||